Pg
1 Home Page
Pg
2 More rocks 3D
Pg
3 Ultraviolation 97 3D
Pg
4 Gemarama 97 3D
Pg
5 Purple Passion 3D
Pg
6 Benitoite 3D
Pg
7 Tenebrescence
Pg
8 Castle Dome 3D
Pg
9 NJESA Show 3D
Pg
10 Uranium Glass 3D
Pg
11 Long Wave 3D
Pg
12 Short Wave 3D
Pg
13 Radioactives
Pg
14 Radioactives
Pg
15 Radioactives
Pg
16 Ruggles Mine 3D
Pg
17 Fluorescent 98 3D
Pg
18 Triboluminescence
Pg
19 Display Case
Pg
20 Franklin Show 3D
Pg
21 Ultraviolation 98 3D
Pg
22 Gemarama 98 3D
Pg
23 Winter 99 3D
Pg
24 Glass
Pg
25 Namibia 3D
Pg
26 Sterling Hill 3D
Pg
27 Sterling Hill
Pg
28 Franklin Dump 3D
Pg
29 Lime Crest 3D
Pg
30 Fluorescent 99 3D
Pg
31 Franklin Dump 2
Pg
32 Ultra 99 3D
Pg
33 Marbles
Pg
34 Delaware 2000 3D
Pg
35 Glo-Glass 3D
Pg
36 Tugtupite 3D
Pg
37 Phila 2000 3D
Pg
38 Belgium 3D
Pg
39 Fluorescent 2000 3D
Pg
40 England 3D
Pg
41 Bancroft
Pg
42 Bancroft2 3D
Pg
43 Crystals 3D
Pg
44 Ultra2001 3D
Pg
45 Cape May
Pg
46 Trotter Dump
Pg
47 West Virginia
Pg
48 Reymert Mine 3D
Pg
49 Ultra2018 3D
Links & Updates
3D Stereo Photography
Fluorescent Minerals
Stereo Home Page
Pg
2 Stereo
Pg
3 Sphalerite
Pg
4 Stereo
Pg
5 Stereo
Pg
6 Stereo
Pg
7 Stereo
Pg
8 Adamite
Pg
9 TerlinguaCalcite
Pg
10 Stereo
Pg
11 Stereo
Pg
12 Castle Dome
Pg
13 Stereo
Pg
14 Radioactives
Pg
15 Stereo
Pg
16 Stereo
Pg
17 Stereo
Pg
18 Stereo
Pg
19 Stereo
Pg
20 Stereo
Radioactive Fluorescent Minerals
Beta Particles
Thorite, Andersonite
Most fluorescent minerals are not radioactive, but some uranium and thorium containing minerals are both slightly radioactive and fluorescent.See 3D Stereo pictures of radioactive minerals
(Click picture to enlarge)
Thorite ( above ) contains the radioactive elements thorium and uranium which each eject alpha particles, not beta, at the first step of each of their decay series. But thorium becomes radium 228 ( at the rate of half in 1.4E10 years ) and the radium 228 ( half-life 5.75 years) emits beta particles. Three of the remaining steps of the series, all with short half-lives, also produce beta. Uranium similarly has a very long half-life but the next two decays in its series are quick and both produce beta.
Beta particles are high speed electrons formed when neutrons decay. They can be stopped by thin plastic shielding.
On this page the Geiger counter was positioned for detecting beta particles in addition to gamma radiation. The detector is calibrated only for gamma but the presence of beta is determined by an increase in the counts when the slotted side of the instrument is aimed at the mineral or other radioactive source. When the Geiger counter is aimed straight at the sample the plastic case blocks the beta particles from reaching the detector tube but there are slots on one side to allow beta detection.
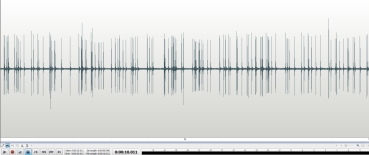
This 10 second sample ( above ) of clicks recorded from the Geiger counter with the beta slots facing the thorite at about one inch shows around 127 counts / 10 sec. or 762 counts / minute.
 |
 |
 |
| Andersonite - Moab, Utah | Andersonite - Moab, Utah, about 1 inch from Geiger counter beta slots | Andersonite - Moab, Utah (shortwave UV) |
Andersonite (above ) Na2CaUO2(CO3)3-6H2O, is a minor ore of uranium. This sample is about 1-1/2" but the andersonite is just a thin coating as can be seen under UV.
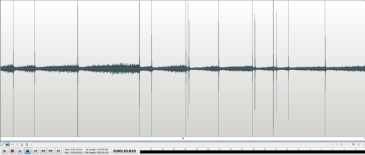
This 10 second sample ( above ) of clicks recorded from the Geiger counter with the beta slots facing the andersonite at about one inch shows around 15 counts / 10 sec. or 90 counts / minute.
James E. Tozour, jtozour@comcast.net
Last Updated 10/9/2015